Great News: Pfizer Vaccine 100% Effective In Tweens, Teens
Pfizer's study found the vaccine is more effective in teens than it is in adults.
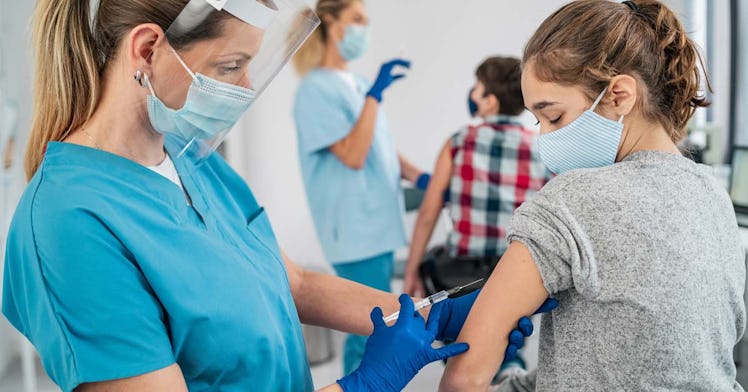
In outstanding news, Pfizer/BioNTech’s clinical trial of kids 12 to 15 has proven that the vaccine is 100 percent effective in preventing illness, and well-tolerated, among that group. The ramifications of the news could mean that the Pfizer vaccine will soon be opened up to kids 12 and up — and could be the tipping point in vaccine eligibility when it comes to fighting to reach herd immunity against COVID-19.
The news comes at a time when general COVID-19 developments are mixed. Yes, more vaccines are making it into people’s arms than ever — and as states open eligibility, there’s reason to believe that Biden’s July 4th mandate — that we’d be grilling with our friends by the time it’s Independence Day — could come true.
On the other hand, AstraZeneca’s vaccine rollout has been plagued by unforced errors and concerns, whether or not founded, over blood clots, and a climbing number of daily cases and lessening of state COVID restrictions has led some experts, including the head of the CDC, to fear we are heading straight into the fourth wave of COVID-19 just as the vaccine output is ramping up in a major way. So, the Pfizer news is well-appreciated.
What to Know About the Vaccine and Teens
The trial, of 2,300 U.S. teens between 12 and 15, suggests that the vaccine is even more effective for teens aged 12 to 15 than it is for adults. That’s pretty huge. The study also found that teens experienced the same vaccine side effects as adults.
Not a single teen who received the vaccine in the Phase 3 Trial got COVID-19, making the Pfizer-BioNTech vaccine 100% effective at preventing illness in teenagers. That’s massive. The study has to be peer-reviewed — and Pfizer and BioNTech will be submitting the data to the FDA to expand emergency authorization of the vaccine to include 12 and 15-year-olds, suggesting that schools and more could actually begin opening more safely than previously expected.
The great news is that the vaccine is so effective in teens, who are known to spread COVID-19 as much as adults.
What Does the Efficacy Mean?
The company will submit the data to Pfizer/BioNTech in order to get emergency authorization to allow kids as young as 12 to begin to be vaccinated with the two-dose vaccine. It’s already been approved in people age 16 and up, so expanding eligibility will be a huge boon for school-aged kids and their parents who are concerned about bringing them back to school in the fall.
“The bottom line is that by the fall I think there’s a good possibility we’ll be vaccinating teenagers, 12 and up, and for middle schools, junior high schools, high schools, it’s really good news in the United States for both teachers and staff. We’ll have teachers and staff vaccinated, we’ll have the students vaccinated in those middle schools and high schools,” said Dr. Peter Hotez of the Texas Children’s Hospital on CNN.
And that’s of course partly what matters: kids can safely return to some semblance of normal life after their vaccination kicks in. They won’t be major spreaders, or victims, of COVID-19, and could even be 100% protected against even getting ill. They will help the U.S. reach herd immunity. It’s all good.
And it’s also important news at a time when the COVID variants and loosened restrictions threaten to overturn the slow and painstaking progress the United States has made in bringing down COVID-19 infection numbers and pushing up vaccinated numbers.
What’s Next?
Pfizer-BioNTech isn’t the only company testing its vaccine on kids and teens. Moderna and Jonson & Johnson immediately began their trials and Pfizer is still testing the vaccine on younger groups. Right now, Pfizer is testing the vaccine on kids age 6 months to 11 years old, working down the age ranges. Next week, it will begin testing on kids ages 2 to 5. Moderna is testing its vaccine on teens and kids in similar age ranges, as is J&J.
While the vaccine probably won’t be available for big kids and younger before the school year starts, the news is still overwhelmingly positive.
This article was originally published on